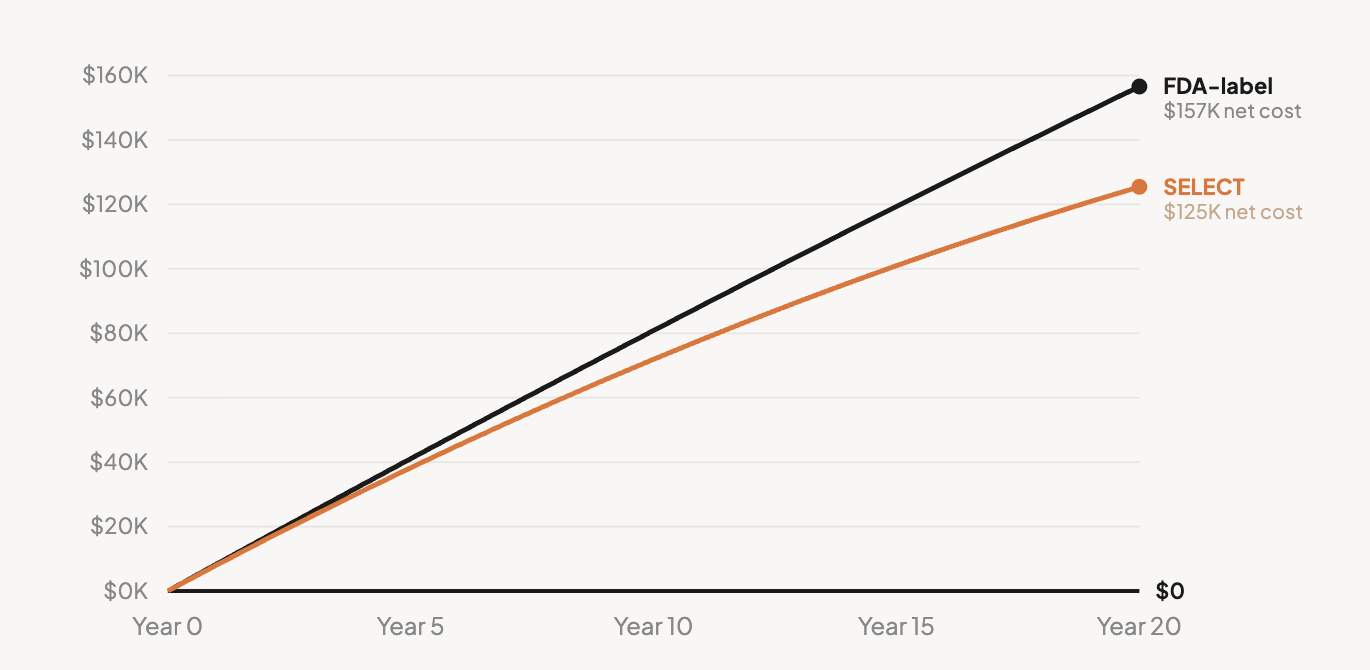
Even under ideal conditions — recommended dose, 100% persistence, the highest-risk patients — broad GLP-1 coverage for obesity doesn't break even on medical savings.
Semaglutide cut major adverse cardiovascular events (MACE) by 20% in SELECT (Lincoff et al., NEJM 2023). Tirzepatide produced up to 22.5% weight loss in SURMOUNT-1 (Jastreboff et al., NEJM 2022). These are large, well-controlled results. The drugs work.
The payer question is different: does broad GLP-1 coverage for obesity produce a positive financial return? We modeled the most drug-favorable scenario possible — every patient at the recommended dose, 100% persistence indefinitely, applied to the highest-risk population with established cardiovascular disease — and the answer is no. The analysis below shows why.
This analysis covers the non-type-2-diabetes population. We model the two benefit pathways with the strongest outcomes evidence — MACE prevention and type 2 diabetes (T2D) prevention — using published trial data and real-world persistence. Where assumptions were required, we chose the option that favors the drug: SELECT's MACE reduction is extrapolated to 20 years (beyond the 3.3-year trial), applied to primary prevention (where no dedicated trial exists), T2D prevention is modeled as permanent (not delayed), and all figures are undiscounted.
The math
We model two populations. They differ in cardiovascular risk, which determines the size of the MACE savings:
| Population | CVD status | Share of insured lives |
|---|---|---|
| SELECT-eligible | Obesity + established cardiovascular disease | ~2% |
| FDA-Label eligible | BMI ≥30 or ≥27 with comorbidity. No CVD required. | ~38% of US adults |
For each population, we model two sets of operating conditions — how patients actually take the drug, which determines how much drug spend accrues relative to the savings:
| Conditions | Dose | Persistence |
|---|---|---|
| Ideal | All patients reach recommended maintenance dose | 100%, indefinitely |
| Real-World | Most patients do not reach recommended maintenance dose | Low persistence at 2 and 3 years |
For Real-World dose, we model 75% of trial benefit — a drug-favorable estimate given that real-world outcomes data consistently show substantially less weight loss than clinical trials. For Real-World persistence, we use approximately 1.9 drug-years per starter over 20 years, derived from Prime Therapeutics data — the most recent published 3-year persistence data available (~63% at 1 year, ~15% at 2 years, 8.1% at 3 years). We examine this structural gap between trial efficacy and real-world effectiveness in The Reality Gap.
For each combination of population and conditions, we compute the medical savings from MACE and T2D prevention over 20 years and compare them to the estimated average employer net GLP-1 drug cost as of May 2026: $700/month, or $8,400/year. The result is the breakeven drug price — the price at which those savings would exactly cover the drug spend. The full methodology shows the component-by-component derivation.
| Population & conditions | Savings per drug-year | Breakeven | Current price | Multiple |
|---|---|---|---|---|
| SELECT — Ideal Recommended dose, 100% persistence | $2,128/yr | $177/mo | $700/mo | 4× |
| SELECT — Real-World Observed dose & persistence | $354/yr | $29/mo | $700/mo | 24× |
| FDA-Label — Ideal Recommended dose, 100% persistence | $571/yr | $48/mo | $700/mo | 15× |
| FDA-Label — Real-World Observed dose & persistence | $83/yr | $7/mo | $700/mo | 100× |
MACE event cost $60K blended. T2D incremental cost $7K/yr. All inputs and derivations in the full methodology.
The best case — SELECT-eligible patients under Ideal conditions that have never been achieved — produces a breakeven of $177/month against a $700/month drug. Under Real-World conditions in the broader coverage population, the drug costs 100× its MACE and T2D savings.
To see why, consider the number needed to treat. In SELECT, the drug cost to prevent one MACE event is roughly $1.85 million. The event itself costs ~$60,000. That ratio — roughly 31 to 1 — is the structural constraint, and it does not improve with better adherence or persistence.
This is why the model includes an Ideal scenario — every patient on the recommended dose, indefinitely. If the math doesn't work there, the constraint is structural, not operational. Better engagement, tighter prior authorization, and improved persistence cannot close a 4× gap in the best case or a 100× gap in the broad population.
For the FDA-Label population under Real-World conditions, the per-patient arithmetic is:
Over 20 years under Ideal conditions, the net cost per starter is $125,440 for SELECT-eligible patients and $156,580 for the FDA-Label population — drug spend minus all modeled medical savings. Under Real-World conditions, discontinuation caps exposure but the gap per drug-year is wider. The full methodology breaks down all four scenarios.
Independent claims data confirms these figures. Prime Therapeutics' analysis across 16.5 million commercially insured members found Year-1 incremental cost per GLP-1 user of $6,994 and Year-2 of $4,206, with two-year cumulative cost of $11,200 (AMCP 2024-2025 data). Further, the benefits that could eventually offset these costs — cardiovascular event reduction, diabetes prevention — accrue over decades, while median employee tenure is five years. The plan paying the drug cost today is rarely the plan that would capture the long-term offset.
What about other benefits?
The breakeven table above reflects savings from MACE and T2D prevention only — the two pathways with the strongest outcomes data. GLP-1s also show clinical efficacy for obstructive sleep apnea, knee osteoarthritis, heart failure, fatty liver disease, and chronic kidney disease, plus cross-sectional associations with improved productivity. None have been monetized in published payer-budget terms. The question is how much they would need to be worth to close the gap:
| If other benefits are worth... | Breakeven rises to... | Gap to $700/mo |
|---|---|---|
| $0/yr (MACE + T2D only) | $7/mo | 100× |
| $1,000/yr | $90/mo | 7.8× |
| $2,500/yr | $215/mo | 3.3× |
| $5,000/yr | $424/mo | 1.7× |
| $8,317/yr Required to justify current pricing | $700/mo | 1.0× |
A Novo Nordisk-funded productivity analysis estimated obesity-attributable costs up to $2,659/yr. High-end OSA care avoidance could add ~$3,000/yr for the ~28% with moderate-to-severe disease. To reach $8,317/yr, every benefit category would need to be at its maximum estimate simultaneously.
What about lower prices?
Oral Wegovy and Foundayo (orforglipron, Eli Lilly's once-daily oral GLP-1 pill) are being offered at roughly $149-$299/month through self-pay channels. At $299/month, the ratio of drug cost to MACE and T2D savings is still 43:1 for the FDA-Label population and 10:1 for SELECT-eligible under Real-World conditions. Only the SELECT Ideal scenario approaches breakeven at oral pricing (1.7:1) — requiring every patient on the recommended dose, indefinitely.
Counterintuitively, improving persistence makes the financial case worse. Each additional drug-year adds $8,400 in cost against $83-$354 in MACE and T2D savings. At current prices, discontinuation is capping the plan's financial exposure.
Benefit, not investment
Broad GLP-1 coverage for obesity is, at current pricing, an employee benefit — not a cost-saving investment. The drugs work. Some plans will cover them for reasons that extend beyond pharmacy-budget arithmetic. But on MACE and T2D prevention — the two strongest evidence-based pathways — the math does not produce a positive return at any plausible price point or time horizon. Independent cost-effectiveness analyses reach a similar conclusion: Hwang et al. (JAMA Health Forum 2025) found semaglutide at $467,676 per quality-adjusted life year and tirzepatide at $197,023/QALY, with 0% probability of cost-effectiveness. The broad ROI case requires high-risk event rates applied to low-risk populations, long-term benefits captured by short-tenure plans, and quality-of-life value counted as medical-claims savings. Until those assumptions hold, broad GLP-1 coverage remains a benefit with a known cost — not an investment with an expected return.
The full model — component breakdowns, all operational scenarios, MACE-rate sensitivity analyses, and the complete assumption set — is in the detailed methodology.
This analysis draws on Key to Health's GLP-1 reference series. Key to Health is an evidence-based behavioral weight management program with a published RCT and 2-year real-world outcomes from 11,365 adults. Learn more.